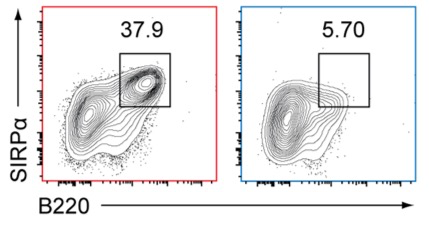
Bcl11a is a transcription factor known to regulate lymphoid and erythroid development. Recent bioinformatic analysis of global gene expression patterns has suggested a role for Bcl11a in the development of dendritic cell (DC) lineages. We tested this hypothesis by analyzing the development of DC and other lineages in Bcl11a (-/-) mice. We found that Bcl11a was required for expression of IL-7 receptor (IL-7R) and Flt3 in early hematopoietic progenitor cells. In addition, we found severely decreased numbers of plasmacytoid dendritic cells (pDCs) in Bcl11a (-/-) fetal livers and in the bone marrow of Bcl11a (-/-) fetal liver chimeras. Moreover, Bcl11a (-/-) cells showed severely impaired in vitro development of Flt3L-derived pDCs and classical DCs (cDCs). In contrast, we found normal in vitro development of DCs from Bcl11a (-/-) fetal liver cells treated with GM-CSF. These results suggest that the persistent cDC development observed in Bcl11a (-/-) fetal liver chimeras reflects derivation from a Bcl11a- and Flt3-independent pathway in vivo.