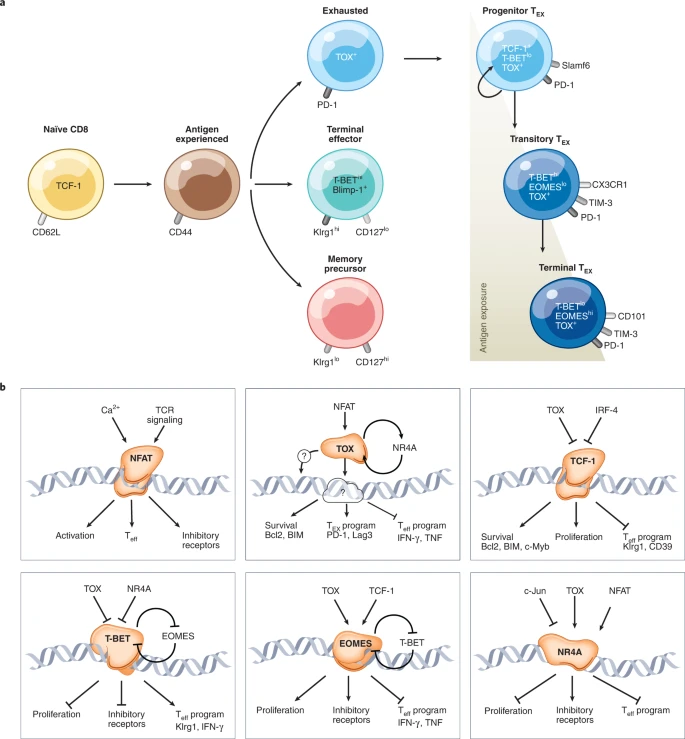
Chronic antigen stimulation during viral infections and cancer can lead to T cell exhaustion, which is characterized by reduced effector function and proliferation, and the expression of inhibitory immune checkpoint receptors. Recent studies have demonstrated that T cell exhaustion results in wholescale epigenetic remodeling that confers phenotypic stability to these cells and prevents T cell reinvigoration by checkpoint blockade. Here, we review foundational technologies to profile the epigenome at multiple scales, including mapping the locations of transcription factors and histone modifications, DNA methylation and three-dimensional genome conformation. We discuss how these technologies have elucidated the development and epigenetic regulation of exhausted T cells and functional implications across viral infection, cancer, autoimmunity and engineered T cell therapies. Finally, we cover emerging multi-omic and genome engineering technologies, current and upcoming opportunities to apply these to T cell exhaustion, and therapeutic opportunities for T cell engineering in the clinic.